Cloud-Based HPC Platform to Support Systemic-Pulmonary Shunting Procedures
Congenital heart diseases (CHDs) account for nearly one-third of all congenital birth defects. Without the ability to alter the prevalence of CHD, interventions and resources must be focused to improve survival and quality of life. The Modified Blalock Taussig Shunt (mBTS) is a common palliative operation on cyanotic heart diseases, but it is associated with significant mortality, the most threatening complications being over-shunting and shunt thrombosis. The proposed application experiment aims at setting up an application to support surgical planning with advanced numerical means, exposed in an interactive and effective manner.
SECTOR: Healthcare
TECHNOLOGY USED: HPC, CFD Simulation
COUNTRY: Italy
The challenge
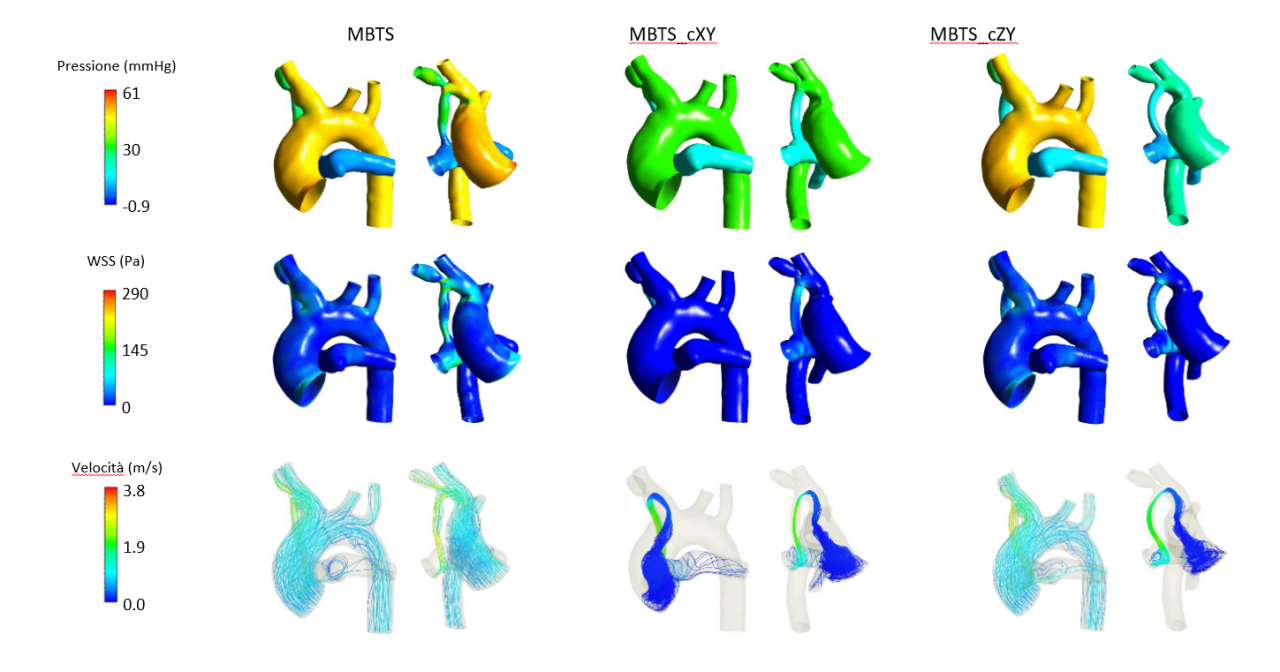
The SMEs InSilicoTrials Technologies and RBF Morph offer a portfolio of software and services to clinics & medical research. To expand its business, the experiment consortium decided to work in the challenging field of Congenital heart disease (CHD). CHDs account for nearly one-third of all congenital birth defects and represented the 7th cause of death in children younger than 1 year in 2017. Without the ability to alter the prevalence of CHD, interventions and resources must be focused on improving survival and quality of life. In this context, the Modified Blalock Taussig Shunt (mBTS) is the most common palliative operation performed. mBTS is used to provide blood flow from the systemic circulation to the lungs. Despite being simple in concept, it is associated with significant morbidity and mortality, the most threatening and lethal complications being over-shunting and shunt thrombosis. Between the risks of shunt thrombosis and over-shunting, the criteria for the definition of the shunt size, length and location, remain a matter of debate. When too small, a shunt diameter would lead to shunting thrombosis due to high wall shear stress (WSS), whereas when too large it may result in a state of pulmonary overflow, decreased diastolic pressure and reduced systemic perfusion. A computational simulation of these effects could support the medical decision, yet this requires substantial know-how and computing power, in order to provide high-quality results and the subsequent surgery in a short timeframe. This experiment creates the opportunity to combine the consortium know-how with fundamental tools supplied by the HPC environment, which combined have the capacity to open new doors for generating business opportunities for the experiment partners in the world of advanced medical modelling.
The solution
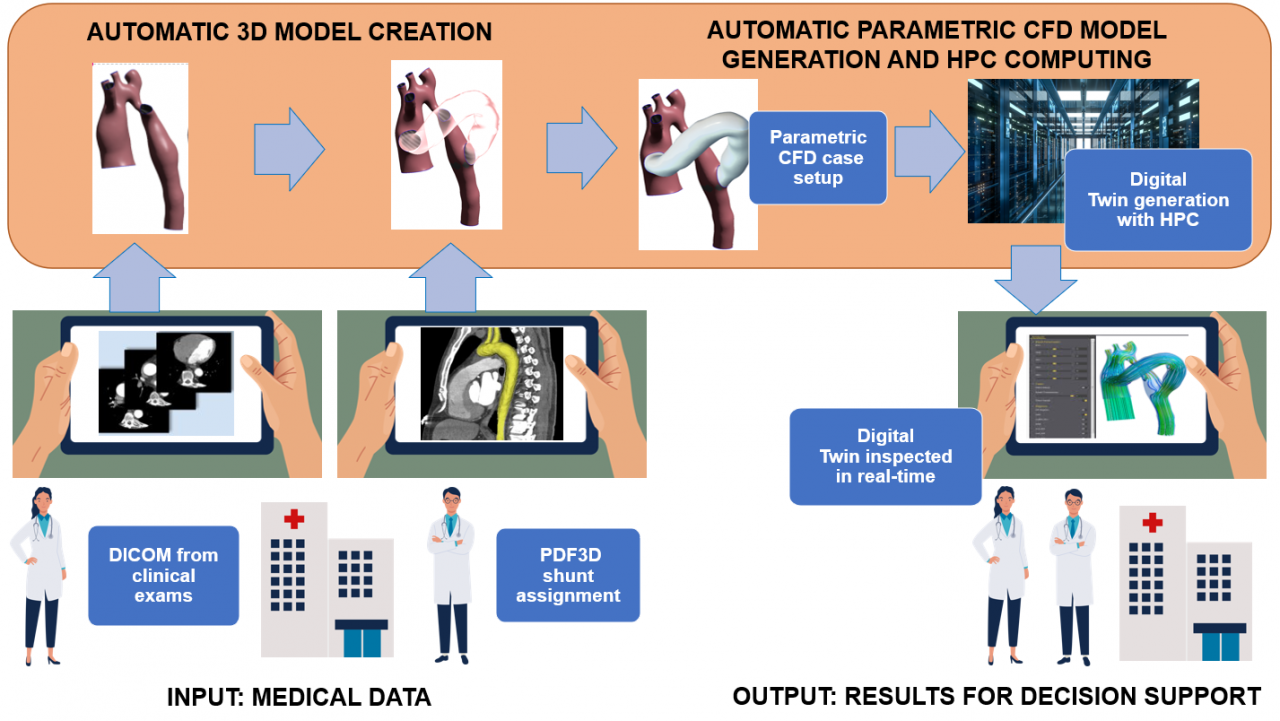
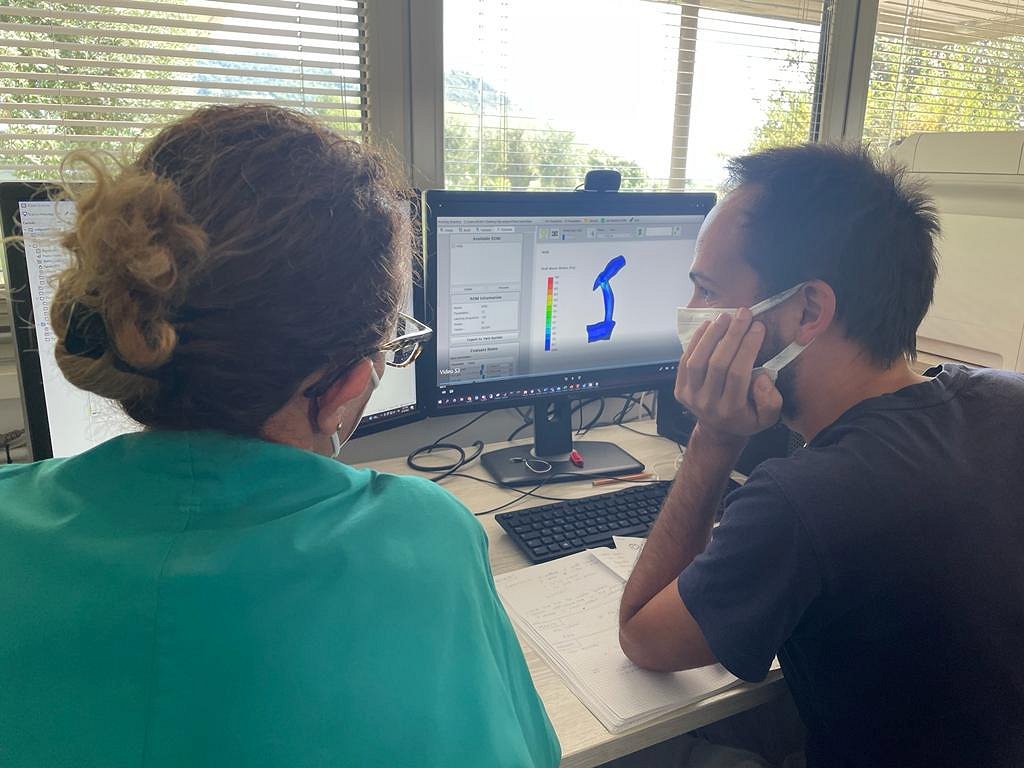
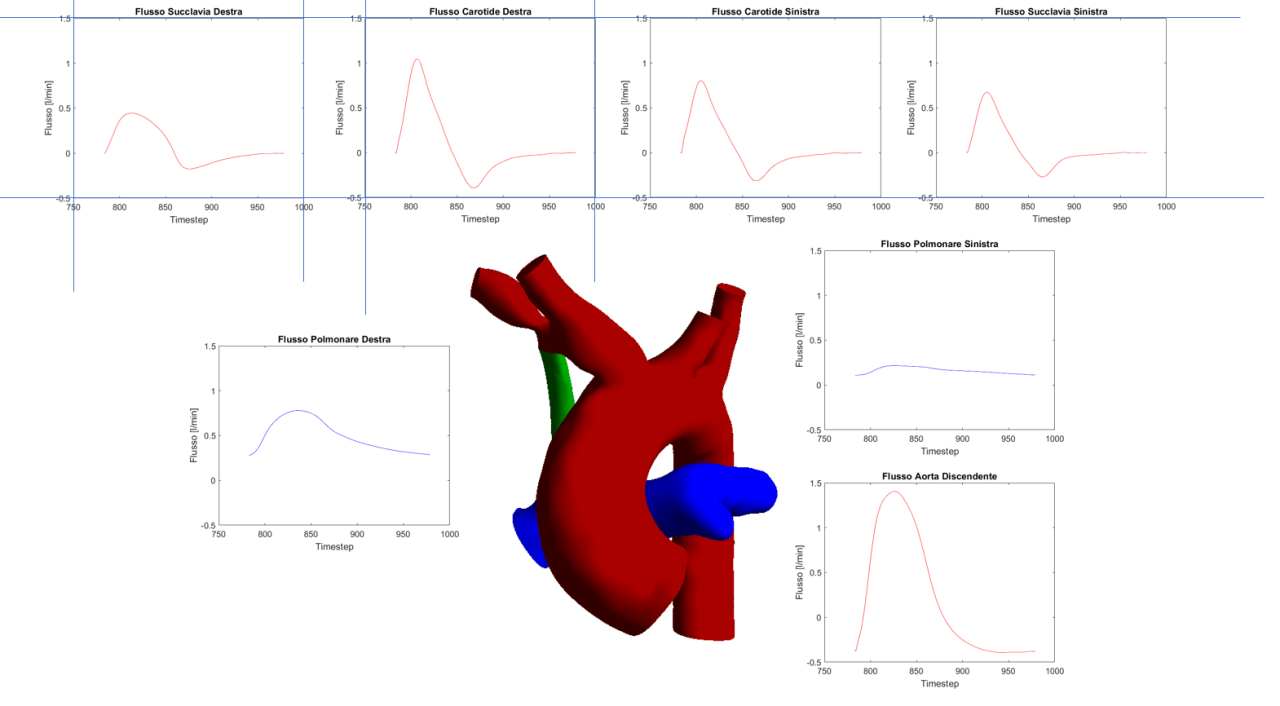
As a solution to the challenge, the experiment generated an affordable decision support web application (“Copernicus”) that, thanks to a “medical digital twin” (MDT), allows surgeons to optimally approach the mBTS medical intervention. Generally, simulating the effect of such a surgery on blood flow requires complex computational fluid dynamics computations on high-performance systems. Typically, effective use of such computations in the field of pre-operative planning is still far from clinical practice, not least because of the specialist know-how needed to run them. Copernicus circumvents these problems by generating the MDT through a Reduced Order Model (ROM) of a patient-specific vascular district, condensing complex and costly pre-loaded computations such that a geometrically parameterized shunt implantation can be used interactively to vary its dimension and positioning. With a dedicated User Interface, the medical staff is thus able to inspect the MDT of the patient and observe how the shunting layout influences the fluid dynamics of the involved impacted area, helping to finalize the decisions on surgical intervention. The use of HPC significantly reduces the time to carry out computing the demanding pre-loaded analysis backing the MDT, a speed-up which is crucial, because important decisions for mBTS preparations must be made in a short time, typically 2 days at worst in the case of complicated morphologies. HPC and the validation through retrospective clinical data moved forward the readiness level of the Copernicus platform, allowing business exploitation with minor additional effort.
Impact
The impact of the solution proposed by Copernicus at the clinical level is highly relevant: The combination of speed and interactivity permits surgeons to find the best treatment option for CDT, thus lowering the incidence of post-surgery complications and reoccurrence, leading to a decrease in the days of hospitalisation of treated patients and related costs. For Fondazione Toscana Gabriele Monasterio, this could mean savings in the order of over €100,000 per year. InSilicoTrials Technologies will add the tool to its portfolio, pursue certification for clinical use, and commercialise it on the market through a SaaS approach, with shares held by RBF Morph and RINA Consulting, and using CINECA’s HPC system as a backend.
In addition, both RBF Morph and RINA Consulting will use the results to increase their presence in the healthcare market: RBF by offering new functionalities in their software library, and RINA Consulting by offering consulting services backed by hardware and software leasing to medical device manufacturers and medical research clinics.
Benefits
InSilicoTrials Technologies: Reach new clients by selling a single service per clinical use after the achievement of software certification (mid-2024), generating an increase in annual turnover of about €450,000 after 4 years.
Clinicians: Improve surgery outcome, reducing hospitalization per patient by 5-6 days on average. In the case of FTGM, savings of more than €100,000 per year are expected.
RBF Morph: Expected increased annual turnover 4 years after the project is €250,000 with the increase of 2 qualified jobs/year.
RINA Consulting: The overall expected increased annual turnover 4 years after the project: €200,000.
CINECA: Additional business as HPC supplier (€30,000 by 2026).
Organizations Involved:
End User: InSilicoTrials Technologies SpA
Technology expert, ISV: RBF Morph Srl
Application expert, End-User: Fondazione Toscana Gabriele Monasterio
Technology expert: RINA Consulting
HPC Centre: CINECA
Partner CINECA is part of the NCC Italy.