HPC for Cancer Drug Accuracy
Presentation of the problem and objective of the experiment
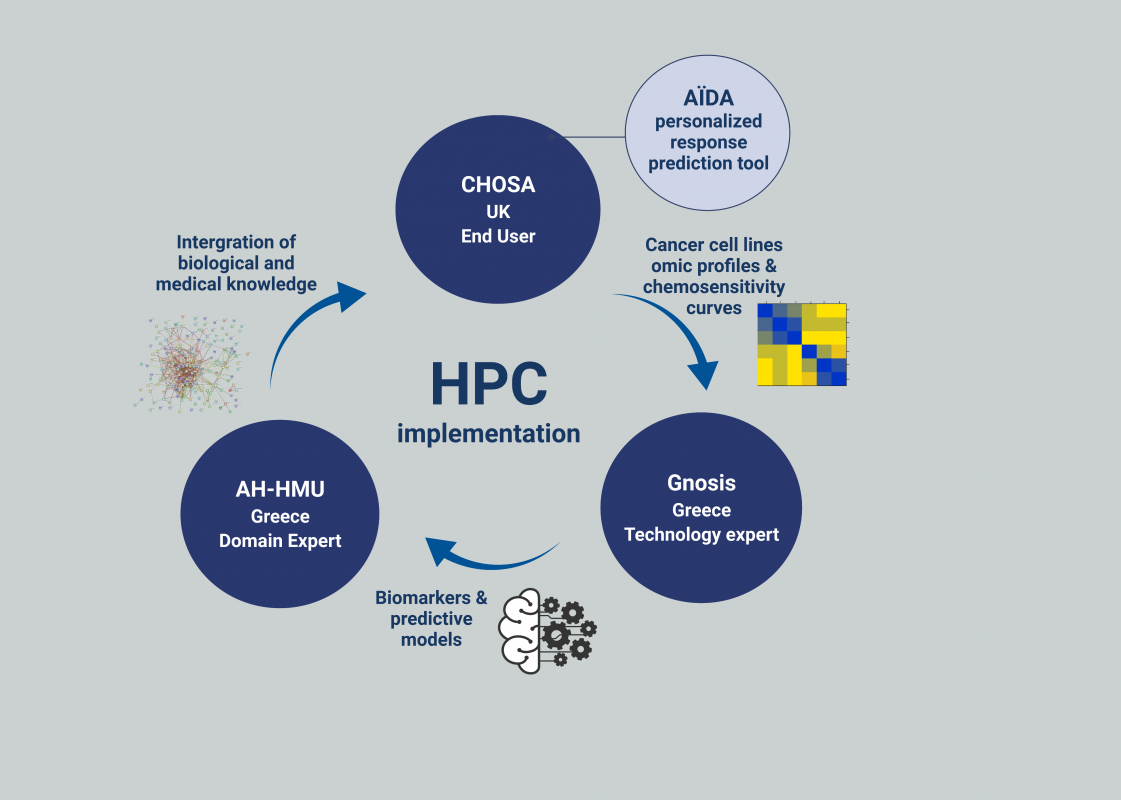
Many cancer patients fail to respond to their drug treatment, resulting in heavy human and economic loss. This lack of efficacy is mainly attributed to host/tumour variations at the genetic and molecular level, which clinical practice still struggles to integrate. The experiment we propose aims to deliver a tangible solution for boosting the development of the personalized response prediction tool Allied Intelligence for Drug Accuracy AÏDA. This tool will consist in an intelligent diagnostic platform able to analyse the molecular profiles of cancer patients and identify the drugs most likely to achieve high effectiveness.
Short description of the experiment
This experiment is based on an extensive analysis of a huge volume of publicly available chemosensitivity and omics data, by using a state-of-the-art AI platform, namely JADBio, developed by one of the partners of the consortium and specialized in automatic machine learning. HPC will play a vital role in the experiment, allowing the execution of experiments that would require a prohibitive amount of time on less performing computational architectures.
UPDATE:
The FF4EuroHPC experiments 1008, HPC for Cancer Drug Accuracy (HPC4CDA), aims to create an intelligent diagnostic platform able to analyse the molecular profiles of cancer patients and identify the drugs most likely to achieve high effectiveness. In this context, partners started the analysis of the transcriptomics profiles of 60 different tumor cell lines, attempting to identify a gene expression signature able to identify tumors highly responsive to the cisplatin compound.
The analyses are performed using JADBio, an AutoML platform that democratizes machine learning for layman life scientists. The first results indicate that predicting the cisplatin effect is feasible up to a reasonable extent. Further analyses for improving this result are currently being carried.
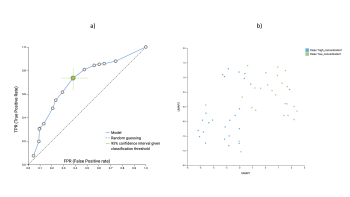
Figure explanation: Initial results from the cisplatin analyses. a) Receiver Operating Characteristic (ROC) curve indicating how well tumors responding to a low concentration of cisplatin can be identified with respect to tumors requiring a higher dose. The Area Under the Curve (AUC) achieved in this analysis is 0.71. b) UMAP represents how tumors requiring only a low concentration (green points) are distributed with respect to tumors requiring higher concentrations (blue points). The two classes are not linearly separable, demonstrating the complexity of the problem at hand.
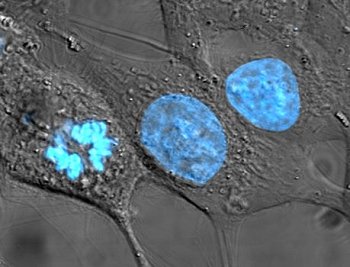
Figure explanation: Cells from the HeLa culture, one of the firsts immortalized cell lines used in cancer research.
HPC for Cancer Drug Accuracy - 10th Santorini Conference.pptx
Organisations involved:
End User: CHOSA Oncology Ltd
Domain Expert: Hellenic Mediterranean University
Technology Expert: JADBio
HPC Provider: Yotta Advanced Computing